
昵称:augmentin warnings 邮箱:Ethefup@mailsphere.xyz
评论于:2025-06-22 18:40:55
The US Food and Drug Administration FDA has granted a 6 month priority review status to the supplemental new drug application filed by the AstraZeneca corporation for the use of anastrozole Arimidex in the adjuvant treatment of early breast cancer in postmenopausal women how to deal with augmentin side effects If you re using a screen reader, or having trouble reading this website, please call Lemonaid Patient Support for help at 888- 536- 2267





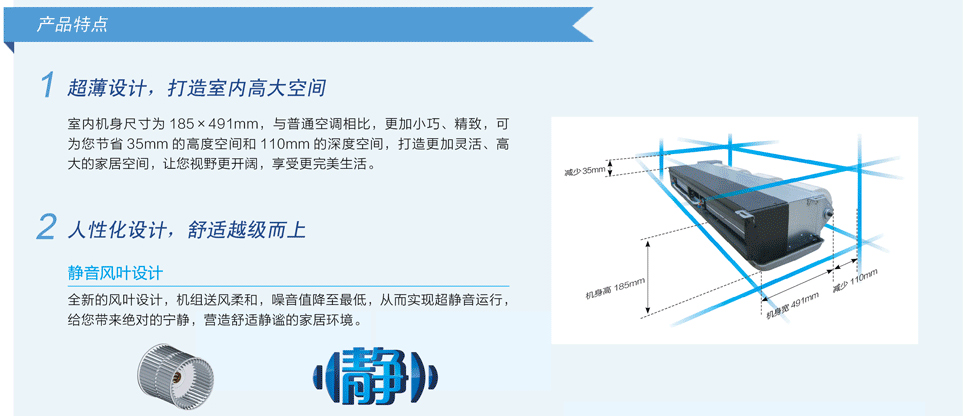







 B系列超薄风管机风口尺寸
B系列超薄风管机风口尺寸
